|
5/21/2023 0 Comments Delta u standard calculator 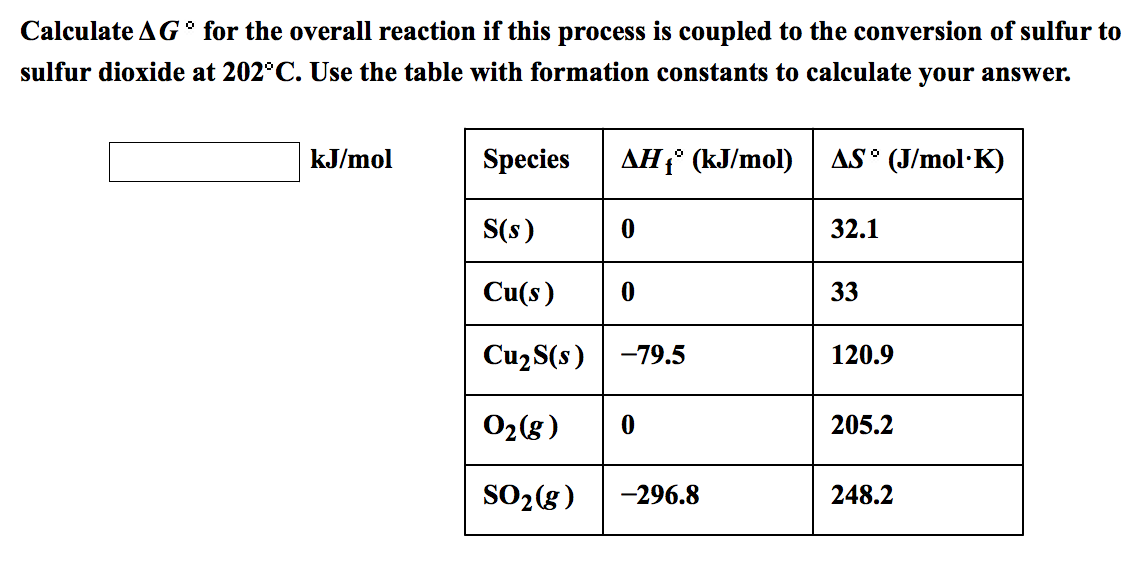
If we know the standardĬhange in free energy for a particular reactionĪt a particular temperature, we can calculate the equilibriumĬonstant for that reaction.  
This equation is extremely useful because now we have a relationship between free energy and equilibrium. If we subtract RT ln of K from both sides, we get delta G naught is equal to negative RT ln of K. And we can plug in K for Q, that gives us zero isĮqual to delta G naught plus RT ln of K. So we can plug in zero forĭelta G in our equation. The reaction quotient Q is equal to the equilibrium constant K. At equilibrium, the instantaneousĭifference in free energy between reactants and products is zero, which means there's no moreĭriving force for the reaction. R is the ideal gas constant, T is the absolute temperature in Kelvin, and Q is the reaction quotient. In their standard states at a specified temperature. This value refers to theĭifference in free energy between reactants and products Standard change in free energy between reactants and products. And from the equation we see that delta G is equal to delta naught plus RT ln of Q. We can think about delta G as the instantaneousĭifference in free energy between reactants and products. Non-standard changes in free energy delta G. First, we need to consider the equation that allows us to calculate And our goal is to thinkĪbout the relationship between free energy and this reaction when it comes to equilibrium. We have a generic reaction where reactants turn into products. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
